
Submitted by ovarian cancer survivor Annie Ellis, OCRA Research Advocate and Scientific Advisory Committee member
This year’s SGO annual meeting was held in Phoenix, Arizona from March 18-21. Approximately 2,100 medical professionals attended in person with several hundred more attending virtually to learn the latest on gynecologic cancers. By now, you may have seen professional coverage of and tweets about this meeting. Below are brief highlights from a research advocate’s perspective.
Biomarkers, Side Effects and Communication
As advances in technology have led to deeper understanding of the complexities of ovarian cancers and more personalized/precision approaches, many research presentations included exploration and analysis of biomarkers to identify subsets of patients who may be most likely, or unlikely, to have their cancers respond to particular treatments or better tolerate treatment at a lower starting dose (predictive biomarkers). Biomarkers can be mutation carrier status (BRCA1 or 2+), molecular characteristics of tumors (HRD, homologous recombination deficiency), ARID1A, “hot” or “cold” indicating presence of T-cells in tumors) or other characteristics such as residual disease after debulking, weight or platelet count. Patients at treatment decision points may have different options based on biomarkers.
What was most exciting to me during this meeting were the many discussions during panels and distillations regarding the importance of communication to educate patients about new treatment options using biomarkers, balance potential risks and potential benefits, and manage anticipated side effects. Some new treatments in clinical trials have side effects that are very different than those with traditional chemotherapy. One presenter emphasized that patients should be provided with a plan to proactively manage side effects and know who to contact between appointments if extra help is needed.
Rare Subtypes
Opening day of the meeting included a four-hour Master Class: Rare but Revelatory: Clinical Updates & Research Advances in Uncommon Gynecologic Cancers. This session featured talks by experts working on rare gynecologic cancers and a panel of patient advocates. With fewer cases diagnosed each year, global efforts are essential to studying rare cancers as well as innovative biostatistical designs, including use of historical controls. Highlights included:
- Lindsay Frazier of Dana Farber Cancer Institute described the efforts of the Malignant Germ Cell International Consortium (MaGIC) in bringing experts together to develop more effective treatments for in germ cell tumors (GCTs). A Patient Advocate Committee has recently been added to MaGIC.
- Rachel Grisham of Memorial Sloan Kettering gave an overview of low grade serous (LGS) Ovarian Cancer and spoke about the work of the International Consortium for Low Grade Serous Ovarian Cancer (ICLC). The ICLC includes LGS survivors and caregivers.
- Stephanie Gaillard and le-Ming Shih from Johns Hopkins Sidney Kimmel Cancer Center presented an overview of clear cell carcinoma and ARID1A mutations and alterations in the P13K/AKT pathway. Tian-Li Wang from Johns Hopkins University presented DNA Methylating Drug Temozolomide Sensitizes ARID1A–Mutated Tumors to PARP Inhibitors.
- Diego Manavella from Yale University presented Ovarian and Uterine Carcinosarcoma Cell Lines Show Preclinical Sensitivity to BAY 1895344, a Novel Ataxia-Telangiectasia and Rad3-related (ATR) Kinase Inhibitor.
- Ovarian cancer survivor and OCRA conference attendee Jane Ludemann from New Zealand shared her story of being diagnosed with low grade serous ovarian cancer and founding Cure Our Ovarian Cancer. Jane is a Steering Committee member of the International Consortium for Low Grade Serous Ovarian Cancer.
- Ovarian cancer survivor and OCRA Research Advocate Kimberly Richardson shared her story of being diagnosed with granulosa cell ovarian cancer. Kimberly created the Survivors Advising Scientists Training Program at the University of Illinois – Chicago (UIC) Cancer Center and is involved in a multitude of advocacy activities.
Additional Rare Subtype presentations at the meeting included:
- During the Late-Breaking Abstract Session, David Gershenson from MD Anderson Cancer Center presented The Genomic Landscape of Low-Grade Serous Ovarian/Peritoneal Carcinoma (LGSOC) and its Impact on Clinical Outcome. LGS patients with tumors that have MAPK mutations had better survival.
- During an education session, Joan Tymon-Rosario presented Homologous Recombination Deficiency (HRD) Signature-3 in Ovarian and Uterine Carcinosarcomas Correlates with Preclinical Sensitivity to Olaparib, a Poly (Adenosine Diphosphate [ADP]- Ribose) Polymerase (PARP) Inhibitor.
- During an education session, Joyce Liu of Dana Farber Cancer Institute presented Activity of Combination Trametinib/Navitoclax in Patients with RAS-mutated Gynecologic (GYN) Cancers in a Phase 1/2 Study. RAS mutations are seen in low grade subtypes including serous, endometrial, and mucinous.
Maintenance
- During an Education Session, Melissa Hardesty of Alaska Womens Cancer Care presented OVARIO, A Phase 2 Study of Niraparib + Bevacizumab in Advanced Ovarian Cancer Following Front-Line Platinum-Based Chemotherapy with Bevacizumab: Updated Analysis. Over 50% of high-risk patients remained progression-free at 24 months and clinical benefit observed across biomarker subgroups. Patients with low weight or low platelets started at a lower dose of niraparib.
- During an Education Session, Joyce Liu of Dana Farber Cancer Institute and Stephanie Wethington of Johns Hopkins discussed “Should All Patients with Primary, Advanced Epithelial Ovarian Cancer Be Offered a PARP Inhibitor?” During this debate, the doctors reviewed clinical trials leading to PARP inhibitor approvals, whether BRCA mutation and HRD status are indicated for specific PARP inhibitors, potential side effects and resistance. Ultimately, both doctors agreed that PARPi maintenance should be offered, but not all patients will accept toxicities in instances where potential benefit appears to be minimal. Communication and education are important so that patients can participate in shared decision-making.
Recurrent Ovarian Cancer
- During the Late-Breaking Abstract Session, Ursula Matulonis from Dana Farber Cancer Institute presented Efficacy and Safety of Mirvetuximab Soravtansine in Patients with Platinum-Resistant Ovarian Cancer with High Folate Receptor Alpha Expression: Results from the SORAYA Study. In this Phase 3 single arm study of 105 patients, 32.4% had a complete (5) or partial response (29). One potential side effect of this Antibody Drug Conjugate is blurred vision which was reversible.
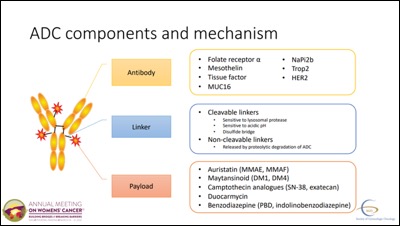
Antibody Drug Conjugates
- During an Education Formum on Integrating Novel Therapeutics into Clinical Practice, Joyce Liu from Dana-Farber Cancer Institute gave a talk on Antibody-Drug Conjugates (ADCs). ADCs target a specific antibody on cancer cells and deliver anti-cancer drugs via a linker.
Survivorship / Nutrition and Diet
The ketogenic diet (KD) is a low-carbohydrate/high-fat diet that is being studied in different cancers to see if KD will help standard treatments work better on cancer cells. However, the results of two studies presented during a plenary session showed that KD promoted tumor growth in ovarian cancer mouse models. It is not known if KD has the same effect in humans, patients on active treatment should discuss any major changes to their diets with the medical teams.
- Mariam Alhilli of the Cleveland Clinic presented Ketogenic Diet Promotes Epithelial Ovarian Cancer Progression and Alters Tumor Gene Expression in-vivo.
- Medical student Surabhi Tewari from the Cleveland Clinic Lerner college of Medicine presented The Impact of Ketogenic Diet on the Gut Microbiome and Tumor Growth in an In Vivo Epithelial Ovarian Cancer Model.
Patient Advocates at SGO
The SGO’s Foundation for Women’s Cancer’s Patient Education Committee (PEC) hosted several activities for patient advocates attending the annual meeting to interact with members of the PEC and ask questions: A Welcome Connection and Poster Walk for those attending in person and a zoom conversation for those attending virtually. Even more activities will be planned for next year’s annual SGO meeting scheduled for March 25-28 in Tampa, Florida.



